Amount of Substance - Balancing Chemical Equations (A-Level Chemistry)
Balancing Equations
Balancing Full Equations
Definitions
The law of conservation of mass states that in a closed system, the mass of the system must remain constant over time and cannot change. This means substances cannot be added or removed from the system. Understanding this fundamental principle is crucial in many scientific and engineering disciplines, and it’s a concept you’ll see applied rigorously during any Chemical Engineering work experience.
A balanced equation is where the number of atoms of each element is the same for both reactants and products. In other words, a balanced equation has the same number of each atom of each element on both sides of the equation. The reason we must have balanced equations is to satisfy the law of conservation of charge and mass
Balancing Equations
To balance an equation, you can only change the number of atoms of each element by adding whole numbers to either the reactants or products.
You can change the number of atoms by changing the coefficient number of each atom (the number in front of the element symbol). You cannot change the number of atoms that follow the element symbol – only the coefficient numbers.
Key Rules for Balancing
1. The number of atoms needs to be the same on both sides. For each element, the number of atoms needs to be the same on each side of the equation for the equation to be balanced. The net charge also needs to be equal if ions are involved.
2. Work element by element. Pick an element, and balance it first. In the process, you may unbalance another element, but don’t stress.
Just focus on the element you have picked and then focus on other elements in the later steps. Eventually, after 2-3 steps you will ‘stumble’ upon a balanced equation.
3. Pick any element first, but avoid sole elements, hydrogen and oxygen. It is best to pick an element in a molecule. Leave sole elements until the end. You should also avoid doing hydrogen and oxygen until the end.
Practice Questions: Balancing Equations
Practice Question: a C3H8 + b O2 —> c H2O + d CO2. Balance the equation to work out the values of a, b, c and d.
1. Firstly write down how many of each atom you have on either side of the equation
Note: LHS = left-hand side
RHS = right-hand side
Carbon: LHS = 3 RHS = 1
Hydrogen: LHS = 8 RHS = 2
Oxygen: LHS = 2 RHS = 3
2. Next use trial and error to balance the equation. Remember to change only the coefficient numbers!
The best way to approach this is to pick one element
Let’s look at Carbon first. We can balance the Carbon easily – d = 3. This leaves us with:
C3H8 + O2 —> H2O + 3CO2.
Carbon: LHS = 3 RHS = 3
Hydrogen: LHS = 8 RHS = 2
Oxygen: LHS = 2 RHS = 7
Now, we can focus on Hydrogen. We should leave the oxygen as on the left it is a stand-alone element, so we can balance it easily at the end.
We can balance the Hydrogen if c = 4. This leaves us with:
C3H8 + O2 —> 4H2O + 3CO2.
Carbon: LHS = 3 RHS = 3
Hydrogen: LHS = 8 RHS = 8
Oxygen: LHS = 2 RHS = 10
Now, we can just put 5 in front of oxygen to fully balance the equation.
Balanced Equation: C3H8 + 5O2 —> 4H2O + 3CO2.
a = 1 b = 5 c = 4 d = 3
Practice Question: Sodium nitrate can react with lead oxide. The unbalanced equation is shown below. What are the values of a, b, c and d?
a NaNO3 + b PbO → c Pb(NO3)2 + d Na2O
Sodium: LHS = 1 RHS = 2
Nitrogen: LHS = 1 RHS = 2
Oxygen: LHS = 3 RHS = 7
Lead: LHS = 1 RHS = 1
We can fix both sodium and nitrogen if a = 2. This also happens to balance the oxygens, making 7 on each side!
Balanced Equation: 2 NaNO3 + PbO → Pb(NO3)2 + Na2O
a = 2 b = 1 c = 1 d = 1
Practice Question: The reaction between sulphuric acid and boric acid produces boron sulphate, with water given off too. The equation is shown below. Balance the equation to work out the values of a, b, c and d.
a H2SO4 + b B(OH)3 → c B2(SO4)3 + d H2O
Hydrogen: LHS = 5 RHS = 2
Sulphur: LHS = 1 RHS = 3
Oxygen: LHS = 7 RHS = 13
Boron: LHS = 1 RHS = 2
We can balance the Boron easily if b = 2. This leaves us with:
Hydrogen: LHS = 8 RHS = 2
Sulphur: LHS = 1 RHS = 3
Oxygen: LHS = 10 RHS = 13
Boron: LHS = 2 RHS = 2
We can balance the sulphur if a = 3. This leaves us with:
Hydrogen: LHS = 12 RHS = 2
Sulphur: LHS = 3 RHS = 3
Oxygen: LHS = 18 RHS = 13
Boron: LHS = 2 RHS = 2
We now need to increase the hydrogen and sulphur on the right. It is best to look at H2O first as the B2(SO4)3 is complicated so we want to avoid it. We can just make d = 6, and everything balances!
Balanced Equation: 3 H2SO4 + 2 B(OH)3 → B2(SO4)3 + 6 H2O
a = 3 b = 2 c = 1 d = 6
Balancing Ionic Equations
Definitions
An ionic equation is an equation for reactions occurring between ions in solution. In an ionic equation the ions are written with their charges, and the symbols of the elements that form the elements.
A spectator ion is an ion that does not change state or charge in the reaction, which means that it does not react. The ion is present in the reaction solution but it does not participate in the reaction
A state symbol is a letter (either s, l, g or aq) that defines the state of matter the compound or molecule is found in.
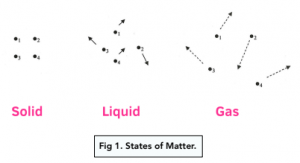
State Symbols
The state symbols that you need to learn are:
( s ) = solid
( l ) = liquid
( g ) = gas
( aq ) = aqueous or in solution or in water
You can’t really work out state symbols. Instead, you’ll start to remember them through memory and general instinct. Here are some tips for working out state symbols when writing equations:
- Metals tend to be insoluble, so tend to be solid (s).
- Acids found in the laboratory tend to be aqueous (aq).
- Elements such as oxygen, hydrogen, nitrogen tend to be gaseous (g).
- If a reaction occurs in solution, normally most components are aqueous (aq).
Practice Question: Work out the state symbols of each reactant and product in this reaction: Mg(s) + H2SO4(aq) → MgSO4(aq) + H2(g)
We will learn later that metal + acid → salt + hydrogen. The metal is very likely to be a solid, as metals are insoluble on their own. The acid will be aqueous, according to our rules. Hopefully you know that hydrogen gas is given off in this reaction (causing the effervescence / bubbling seen), so we can confidently say hydrogen is a gas. And a salt is formed, MgSO4. Most salts are in solution, so magnesium sulphate is likely to be aqueous.
Ionic Equations
- In an ionic equation, spectator ions are removed. Spectator ions are removed from an ionic equation because they do no participate in the reaction. An ionic equation only includes particles that participate in the reaction.
- The products formed by reacting particles is also included in an ionic equation. In an ionic equation both particles and products are included as long as they are reacting in the solution.
- An ionic equation must also be balanced. An ionic equation is treated exactly the same as a full equation meaning the number of ions on both sides of the equation must equal.

Practice Questions: Balancing Ionic Equations
Practice Question: If the full equation of the reaction between zinc metal and copper sulphate is Zn (s) + CuSO4 (aq) –> ZnSO4 (aq) + Cu (s), what is the ionic equation?
1. First of all write out a complete ionic equation by splitting compounds into their constituent ions.
E.g. CuSO4 is a compound that is made up of the ions Cu2+ and SO42-. We split the compounds into their ions as when the reaction takes place in solution, the reactants will dissolve and break into ions.
Zn (s) + Cu2+ (aq) + SO42- (aq) —> Zn2+ (aq) + SO42- (aq) + Cu (s)
2. Next remove spectator ions. These are ions that do not change state and so do not participate in the reaction.
There is only one spectator ion in this equation which is SO42- (aq) because it is present on either side of the equation, but it does not change state. We therefore remove SO42- (aq) from either side of the equation.
Zn (s) + Cu2+ (aq) —> Zn2+ (aq) + Cu (s)
3. Finally write out the net ionic equation which only includes reacting particles or in other words the spectator ions are removed.
Ensure the ionic equation is balanced both in terms of number of atoms of each element as well as the the charges on either side of the equation.
Zn (s) + Cu2+ (aq) —> Zn2+ (aq) + Cu (s)
4. Check that the equations is balanced and the charges are equal on both sides.
In this equation the numbers of atoms of each element on either side is the same and the charge on either side of the equation is 2+.
The amount of substance is a measure of the quantity of matter present in a substance. It is usually measured in moles (mol) and is used to balance chemical equations in A-Level Chemistry.
Moles are used to balance chemical equations because they provide a consistent way of measuring the amount of each substance involved in a reaction. By using moles, we can ensure that the number of atoms or molecules of each substance is the same on both sides of the equation, which is necessary for a chemical reaction to occur.
The number of moles in a substance can be calculated using the formula: moles = mass ÷ molar mass. The molar mass is the mass of one mole of a substance and can be found in the periodic table.
Balancing chemical equations is important because it helps us understand the relationships between the reactants and products in a chemical reaction. By balancing the equation, we can determine the mole ratios of the reactants and products and predict the outcome of the reaction. Additionally, balanced chemical equations are necessary for accurate calculations of reaction yields, energy changes, and other important parameters in chemistry.
To balance a chemical equation, you need to make sure that the number of atoms of each element is the same on both sides of the equation. This can be done by adding coefficients in front of the formulas of the reactants and products until the number of atoms of each element is equal on both sides. It’s also important to keep in mind the laws of conservation of mass and charge as you balance the equation.
Sure! Let’s take the reaction between hydrogen gas (H2) and oxygen gas (O2) to form water (H2O). To balance the equation, we start by writing the unbalanced equation: H2 + O2 -> H2O. Then, we add a coefficient of 2 in front of the O2 to balance the number of oxygen atoms on both sides: H2 + 2O2 -> 2H2O. The balanced equation is now: H2 + O2 -> H2O.





Still got a question? Leave a comment
Leave a comment