Acids and Bases - More Ka Calculations (A-Level Chemistry)
More Ka Calculations
The Acid Dissociation Constant
Using Ka to work out the Concentration of Weak Acids
The expression for Ka can also be used to determine the initial concentration of a weak acid, given its pH.
Worked Example: Calculate the concentration of a solution of pH = 4.88 of ethanoic acid at 298 K. The value of Ka for ethanoic acid at this temperature = ![]()
Answer:
1. Use the pH value to find the [H+]
![Use the pH value to find the [H+]](https://studymind.co.uk/wp-content/uploads/2022/04/2-84.png)
2. Substitute the value for H+ into the expression for Ka and rearrange.
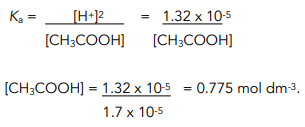
Using Experimental Data to calculate Ka
Previously we looked at how Ka can be used to calculate the pH of a weak acid. By working backwards, we can calculate the Ka for an acid from its pH and a known mass.
Worked Example: The pH of a solution of ethanoic acid (CH3COOH), made by dissolving 1.31g of ethanoic acid in 250cm³ of distilled water, was found to be 2.84. Calculate the value of Ka for the acid. Ar values: C = 12.0, O = 16.0, H = 1.0
Answer:
1. Find the number of moles of ethanoic acid in the solution.
Moles = mass / Mr
Moles = 1.31g / [(2×12.0) + (2×16.0) + (4×1.0)] = 0.02183… mol
2. Calculate the concentration of the acid solution
Concentration = moles / volume
Volume = 250 cm³ = 0.25 dm³
Concentration = 0.02183 / 0.25 dm³ = 0.0873… mol dm³
3. Unlog the pH to find the [H+]
![Unlog the pH to find the [H+]](https://studymind.co.uk/wp-content/uploads/2022/04/4-57.png)
4. Substitute equilibrium concentrations into the Ka equation.
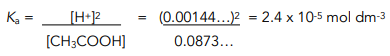





Still got a question? Leave a comment
Leave a comment