Rate Equations - Uses of Clock Reactions (A-Level Chemistry)
Uses of Clock Reactions
Clock Reactions
A clock reaction is a simplified version of the traditional initial rates method to determine orders of reaction.
A clock reaction involves measuring the time taken for a particular amount of product to be produced by a chemical reaction for different reactant concentrations.
An observable end point is used to determine when the wanted amount of product has been produced and hence when to stop the timer.
Product concentration will suddenly increase when the limiting reactant is used up.
When carrying out a clock reaction, we assume that:
- Changes in reactant concentration are insignificant
- Temperature does not change
- The end point is reached before the reaction has gone too far
The Iodine Clock Reaction
1.Iodine is produced. Sulfuric acid is mixed with potassium iodide solution to produce a mixture of H+ and I- ions. Excess hydrogen peroxide is added to the mixture so that iodine is produced by the reaction below:
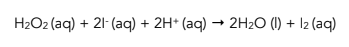
2. Starch solution added. Starch is an indicator that turns dark blue in the presence of iodine.
3. Sodium thiosulfate is added. At the same time you start the stopwatch, sodium thiosulfate solution is added to the reaction mixture. The thiosulfate ions initially react with the iodine formed by the above reaction as shown by the reaction below. The mixture turns colourless as the iodine gets all used up.
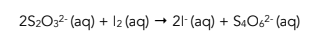
4. The end point is reached. As the thiosulfate ions get used up, any iodine formed by the first reaction stops being used up and instead stays in solution. As a result, the reaction mixture turns dark blue again. This is the end point of the reaction and the stopwatch is stopped.
5. Repeat the reaction. The experiment is then repeated for different concentrations of potassium iodide or hydrogen peroxide. The time taken for the end point to be reached is recorded each time and used to calculate initial rate of reaction with respect to one of these reactants.
FAQs
A rate equation in chemistry is a mathematical expression that describes the relationship between the rate of a chemical reaction and the concentrations of the reactants. It helps chemists understand how changing the concentration of reactants affects the rate of the reaction.
A clock reaction is a type of chemical reaction that can be used to measure the rate of a reaction. These reactions have a visible change, such as a color change, that occurs at a predictable rate. By measuring the time it takes for the visible change to occur, chemists can calculate the rate of the reaction.
Clock reactions have a number of uses in chemistry, including:
Determining the rate equation for a reaction: By performing a clock reaction under various conditions and measuring the rate of the reaction, chemists can determine the rate equation for the reaction.
Studying reaction kinetics: Clock reactions can be used to study the kinetics of a reaction, including the factors that affect the rate of the reaction and the mechanism of the reaction.
Investigating the effect of inhibitors on a reaction: By adding inhibitors to a clock reaction and measuring the rate of the reaction, chemists can investigate the effect of the inhibitors on the reaction.
Testing the efficacy of catalysts: Clock reactions can be used to test the efficacy of catalysts, which are substances that speed up the rate of a reaction.
Clock reactions are performed in the laboratory by mixing the reactants in a flask or test tube and measuring the time it takes for the visible change to occur. The reaction mixture can be stirred or shaken to ensure that the reactants are mixed thoroughly.
The rate of a clock reaction is determined by measuring the time it takes for the visible change to occur. This can be done using a stopwatch or a timing device such as a spectrophotometer. The rate of the reaction is then calculated based on the amount of reactants consumed over time.
There are some limitations to using clock reactions, including:
Complex reaction mechanisms: Clock reactions are most useful when the reaction mechanism is simple and well understood. If the mechanism is complex, it can be difficult to determine the rate equation for the reaction using a clock reaction.
Interference from other reactions: Clock reactions can be affected by other reactions occurring in the reaction mixture, which can interfere with the measurement of the rate of the reaction.
Reactant consumption: Clock reactions rely on the consumption of reactants to measure the rate of the reaction. If the reactants are not consumed at a predictable rate, it can be difficult to determine the rate of the reaction accurately.






Still got a question? Leave a comment
Leave a comment