Amounts of Substances (GCSE Chemistry)
Amounts of Substances
Using Balanced Equations to Work Out Reacting Masses
If we have a balanced equation, we can see the number of moles that are present. If we want to find the mass of one of the molecules, we can use a balanced equation to help us.
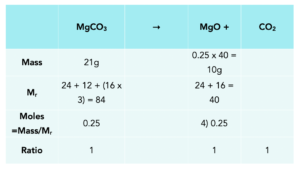
Practice Question: Magnesium carbonate breaks down when heated to produce magnesium oxide and carbon dioxide gas.
MgCO3(s) → MgO(s) + CO2(g)
A student heats 21g of magnesium carbonate strongly in a crucible.
Calculate the mass of magnesium oxide produced by the reaction.
The table below is a simple method to help you with answering these questions. When you move Down the table you Divide, up the table you multiply.
1.Fill in the known values. In our table we know the mass of MgCO3, the Mr of MgCO3 and MgO.
2. Calculate the moles of MgCO3. This is the only molecule which has known values, which is 21g of MgCO
moles = mass/Mr
3. Work out the ratio of moles. We can take this from the equation. We look at the numbers in front of our molecules, the multipliers. In this equation all the multipliers are 1, so the ratio is 1:1:1
4. Look for equal moles. Now we know that the moles of MgCO3 and MgO are the same, as the ratio is equal, so we can add 0.25 moles to the table.
5. Work out the mass of MgO. We can work out the mass of MgO using the moles and Mr which we have worked out.
Rearrange the formula to give: mass = moles x Mr
Therefore, the mass of MgO produced in this equation is 10g.
Experiment to Determine the Empirical Formula of a Metal Oxide
We can do experiments to determine to empirical formula of a metal oxide, either by combustion or by reduction.
Combustion
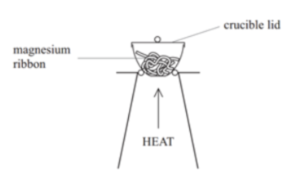
- Take your initial measurements. Weigh the crucible with the lid, then repeat but with the magnesium ribbon added.
- Work out the mass of the magnesium. Find the difference between the two readings in step one to determine the mass of the magnesium.
- Heat the crucible. With the lid on, heat the crucible using a bunsen burner until the magnesium burns. Occasionally lift the lid of the crucible to allow a small amount of air to enter.
- Stop heating when the reaction stops. When the magnesium stops burning, turn off the bunsen burner.
- Reweigh the crucible. Let the crucible cool down, then reweigh.
- Repeat the experiment. Repeat until the mass of the crucible no longer changes, meaning that all the magnesium has burned.
- Calculate the mass of magnesium oxide formed. Find the difference between the mass after heating and the mass of the crucible to find the mass of the magnesium oxide.
- Calculate the empirical formula. Work out the empirical formula of the magnesium oxide, using the mass of magnesium oxide recorded.
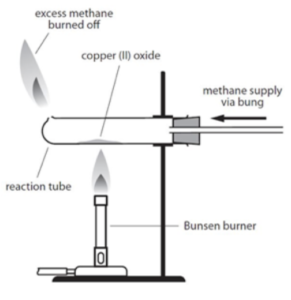
Reduction
- Set up the apparatus. Set up the apparatus as shown in the diagram below. The test tube should be horizontal.





Still got a question? Leave a comment
Leave a comment