The Phosphorus Cycle (A-level Biology)
The Phosphorus Cycle
Phosphorus Cycle
Overview of the Phosphorus Cycle
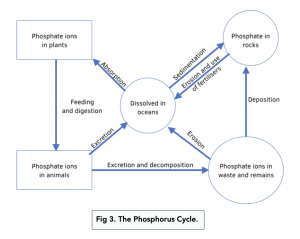
The phosphorus cycle shows how phosphate ions are transferred through an ecosystem.
- Phosphate ions are released. As the sedimentary rock erodes, the ions are transferred into soil, sea, lakes and rivers.
- Plants absorb the phosphate ions through their roots. Both land and aquatic producers absorb the ions, assisted by mycorrhizae, which increases the rate of absorption. The plants use the phosphate ions to make things like phospholipids and ATP.
- Consumers eat plants to obtain the phosphate materials.
- Organic matter is broken down by saprobionic bacteria. The decomposition of dead plants, animals and waste releases phosphate ions back to the soil.
Lost Nutrients
When plants are harvested, the minerals they absorbed from the soil are not replaced by the natural process of decomposition. This leads to soil that is not nutrient rich and will not encourage growth.
The same issue follows removing livestock from fields or slaughtering animals for consumption. The animals eat grass and organic matter, and these nutrients are not replaced through the decomposition of their remains or waste.
Fertiliser is necessary in these cases to replace lost minerals.
- Natural fertilisers are organic material – such as applying dead plants, animals or waste to the soil. The decomposition of the material leads to ions being replaced.
- Artificial fertilisers are inorganic material – they are concentrated solutions of the lost minerals that are sprayed onto the plants.
Artificial fertilizers are faster but have higher environmental risks.
Leaching
Leaching is where fertilisers are washed into rivers and lakes. This occurs when too much fertiliser is applied, or during heavy rainfall, removing the water soluble compounds from the soil.
Artificial fertilisers cause a greater risk for leaching. Natural fertilisers allow for a more controlled uptake of minerals as they are still contained within organic molecules.
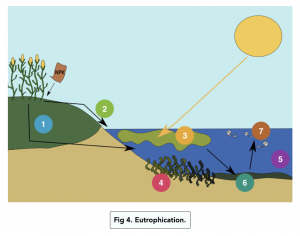
Eutrophication
- Chemical fertilisers leach into water sources. Nitrates are soluble and are readily leached into ponds and lakes.
- The fertiliser accumulates, and is absorbed and utilised by algae. The excess of nitrates will lead to an algae bloom (a rapidly increasing population of algae).
- Light is unable to penetrate past the algae. Algae grows rapidly on the surface of the water, blocking any light.
- Plants cannot photosynthesise and will die.
- Animal species diversity will decrease. Many organisms rely on the plants for food and shelter, so when these die it disrupts the food chain.
- Respiration of decomposers uses up oxygen in the water. Dead organic matter provides more nutrients to the saprobionic bacteria. The bacteria respire aerobically, so use up the oxygen.
- This causes fish and other aquatic organisms to die.
The phosphorus cycle is the movement of phosphorus through the environment, including the biosphere, lithosphere, hydrosphere, and atmosphere. This cycle is important because phosphorus is an essential nutrient for all living organisms.
The phosphorus cycle works through a series of processes, including:
Phosphorus uptake by plants: Phosphorus is taken up by plants through their roots and used to build molecules such as DNA, RNA, and ATP.
Decomposition of dead plants and animals: When plants and animals die, their remains are decomposed by decomposers such as bacteria and fungi, releasing phosphorus back into the soil.
Soil phosphorus: Some of the phosphorus released during decomposition is taken up by plants and reused, while some of it is lost from the soil through runoff and leaching.
Phosphorus in water: Phosphorus in water can be taken up by aquatic plants and animals, or it can be lost from the water through runoff, leaching, and precipitation.
Phosphorus in the atmosphere: Phosphorus in the atmosphere is primarily in the form of phosphates in aerosols, which can be transported long distances before being deposited on land or in water.
The phosphorus cycle is important because it helps to regulate the availability of phosphorus in the environment. This is important because phosphorus is an essential nutrient for all living organisms, and without an adequate supply of phosphorus, growth and reproduction would be limited.
Human activity can impact the phosphorus cycle in several ways, including:
Fertilizer use: The use of fertilizers can increase the amount of phosphorus in the soil, leading to over-fertilization and runoff into water bodies.
Land use changes: Changes in land use, such as deforestation and urbanization, can reduce the amount of phosphorus in the soil and increase the amount of phosphorus in runoff and leaching.
Sewage discharge: Sewage discharge can increase the amount of phosphorus in water bodies, leading to eutrophication and changes in aquatic ecosystems.
The impacts of human activity on the phosphorus cycle can be mitigated through a variety of strategies, including:
Improved fertilizer management: This can help to reduce the amount of phosphorus in runoff and leaching.
Conservation of natural areas: This can help to maintain the natural processes of the phosphorus cycle and reduce the amount of phosphorus in runoff and leaching.
Treatment of sewage: This can help to reduce the amount of phosphorus in water bodies.
Eutrophication is the process by which a body of water becomes enriched with nutrients, leading to excessive growth of algae and other aquatic plants. This can have negative impacts on aquatic ecosystems, including reduced oxygen levels, changes in species composition, and increased disease outbreaks. Understanding eutrophication is important in relation to the phosphorus cycle because it highlights the impacts of human activity on the availability of phosphorus in the environment and the need to manage this resource sustainably.






Still got a question? Leave a comment
Leave a comment