Practice Model of Water - 3.2.1 Internal Energy (GCSE Physics AQA)
Internal Energy
Energy of Particles
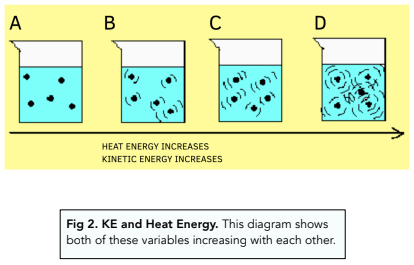
- Particles have kinetic (KE) and potential (PE) energy. In a system, particles mainly have 2 different types of energy. Kinetic energy allows the particles to move. Potential energy is the energy that keeps the particles in their positions.
- Internal energy is the total KE and PE. In the system, if you combine all the kinetic energy and potential energy, and this will give you a value for the internal energy.
Internal energy is the total kinetic energy and potential energy of all the particles (atoms and molecules) that make up a system.
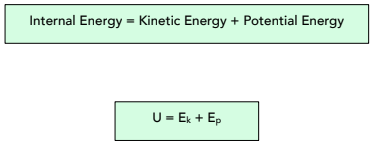
Where:
- internal Energy (U) = Joules, J
- kinetic Energy (Ek) = Joules, J
- potential Energy (Ep) = Joules, J
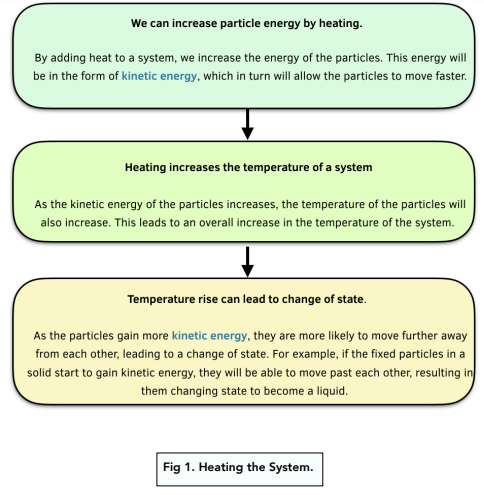
Heating the System





Still got a question? Leave a comment
Leave a comment